As a molecular researcher, it seems all too common to find yourself dealing with the challenges of PCR amplification, especially when working with complex samples that contain GC-rich content, high NaCl content, or strong inhibitors such as EDTA, heparin, or blood. Conventional DNA polymerases like Taq and even designer ones like Phusion and Q5 often struggle to perform optimally under these difficult conditions, leading to suboptimal amplification, unspecific amplification, or even complete failure. So let's discuss the need for a designer ultra-fidelity polymerase based on next-generation fusion technology that can overcome these challenges and enable efficient and accurate amplification even in the most demanding situations.
Challenges with Conventional Polymerases
1. GC-rich content
GC-rich regions in DNA are known for their propensity to form stable secondary structures, which can obstruct the progress of DNA polymerases during amplification. Conventional polymerases often struggle to amplify GC-rich templates, resulting in poor yields and sequence errors. An ultra-fidelity polymerase with enhanced processivity and the ability to disrupt these secondary structures would be invaluable in overcoming this challenge.
2. High NaCl content
High salt concentrations can inhibit the activity of DNA polymerases by interfering with the enzyme's binding to its substrate. The presence of high NaCl content in a sample can reduce the efficiency of PCR amplification, leading to suboptimal results. A designer ultra-fidelity polymerase with increased tolerance to high salt concentrations would allow researchers to obtain reliable results from samples with high NaCl content.
3. Presence of strong inhibitors
Inhibitors such as EDTA, heparin, and blood can severely impede the activity of DNA polymerases. These inhibitors can be present in samples due to sample collection, storage, or extraction methods. Conventional polymerases often struggle to perform efficiently in the presence of these inhibitors, leading to poor amplification and increased nonspecific amplification. A next-generation ultra-fidelity polymerase with improved inhibitor tolerance would greatly enhance the success rate of PCR amplification from such samples.
Advantages of an Ultra-Fidelity Polymerase for Extreme Specificity
In addition to overcoming the challenges presented by complex samples, a designer ultra-fidelity polymerase would also be highly beneficial in situations where extreme specificity is required, such as in the amplification of small amplicons from human genomic DNA. These small amplicons can be problematic due to their high sequence similarity to other regions in the genome, leading to nonspecific amplification and high background noise. An ultra-fidelity polymerase with enhanced specificity and reduced error rates would significantly improve the accuracy and reliability of amplification in these scenarios.
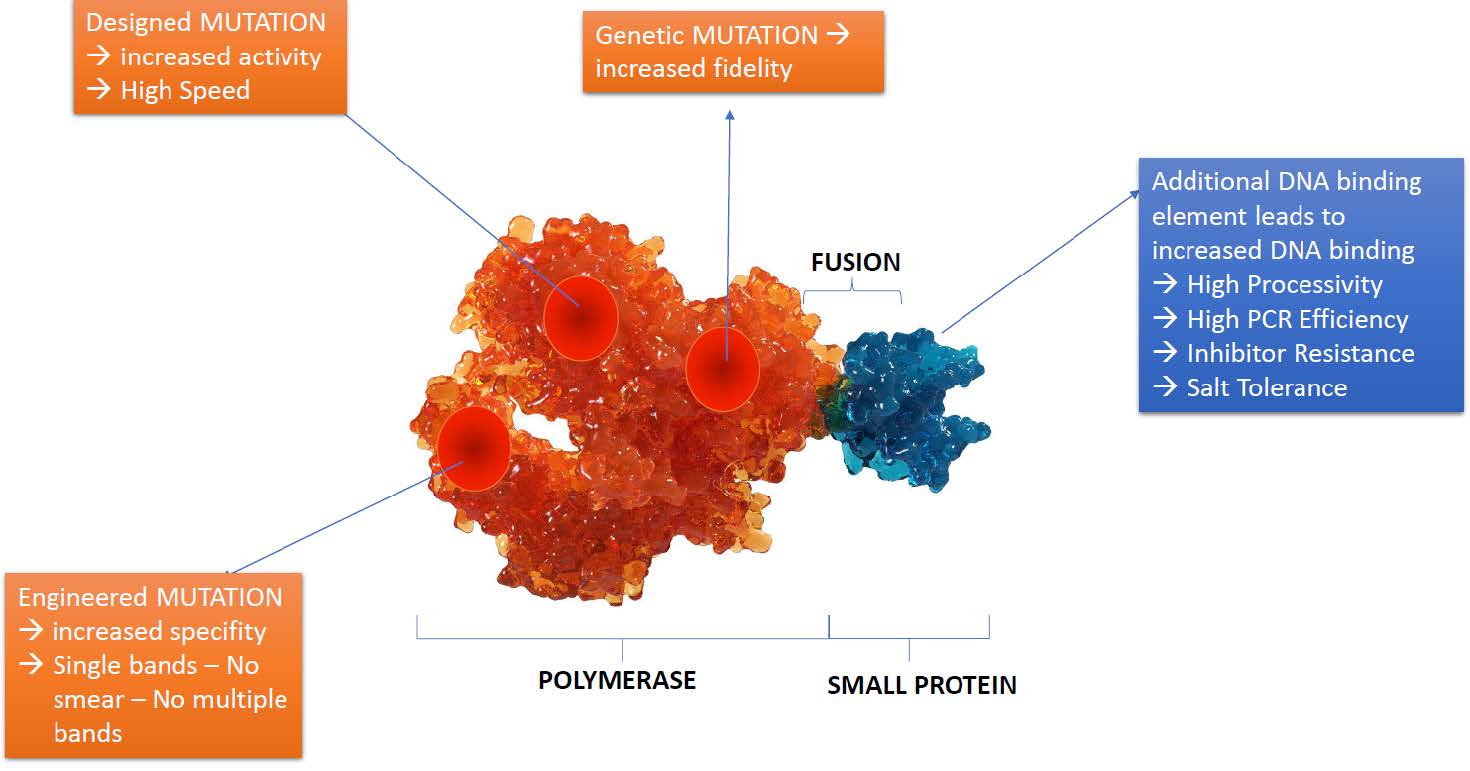
Next-Generation Fusion Polymerase Technology
The development of a designer ultra-fidelity polymerase relies on next-generation fusion polymerase technology. Fusion polymerases combine the desirable properties of multiple enzymes, such as high processivity, high fidelity, and enhanced inhibitor tolerance, to create a single enzyme with superior performance characteristics. By carefully selecting and fusing the appropriate enzymes, it's possible to create a polymerase with the desired attributes to tackle the challenges presented by complex samples and extreme specificity requirements.
The need for a designer ultra-fidelity polymerase that can overcome the challenges associated with complex samples and extreme specificity requirements is clear. The development of such a polymerase, based on next-generation fusion technology, greatly enhances the success and reliability of PCR amplification in molecular research. Not only does this enable researchers to obtain more accurate and reliable results, but it also significantly reduces the time and resources spent troubleshooting and optimizing PCR conditions.
Contact us if you'd like to try a FREE kit of the Indominus™ Ultra-Fi PCR Kit? Also, check out the Instructions for Use!



Deixar comentário
Este site é protegido por hCaptcha e a Política de privacidade e os Termos de serviço do hCaptcha se aplicam.